Correct regulation of the major carbohydrate pathways glycolysis and gluconeogenesis is
essential for organisms. Their defective regulation leads to diabetes in humans. When glucose
becomes available catabolite degradation of the regulatory gluconeogenic enzymes
fructose-1,6-bisphosphatase, phosphoenolpyruvate carboxykinase and cytoplasmic malate dehydrogenase
by the ubiquitin-proteasome-system is a central regulatory mechanism in the model eukaryote yeast.
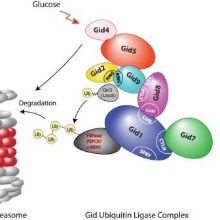
The publication for the first time describes the topology of the Gid ubiquitin ligase complex which
consists of seven different subunits. This RING-ubiquitin ligase complex triggers
polyubiquitination of the gluconeogenic enzymes and by this their proteasomal degradation. The
interaction between the subunits is governed by remarkable domains.