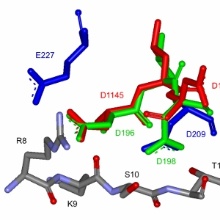
In this article we investigate the target peptide recogniton and methlyaiton by the human SUV39H2 protein lysine methyltransferase. We show that the enzyme recognizes a long sequence on histone H3 comprising A7-G13 and could not identify additional substrates besides histone H3. The substrate sequence preferences could be explained on the basis of the structure of the enzyme. Kinetic analyses show that SUV39H2 prefers H3K9me0 as substrate and it processively introduces two methyl group. The N324K SUV39H2 mutant identified in a genetic disease of the nasal epithelium in dogs is inactive and structurally changed indicating that H3K9 mehtlyaiton by SUV39H2 plays an improtant role in the developmen of the nasal epithelium.